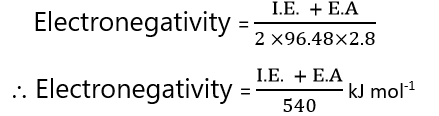Mulliken defined the electronegativity of an atom as the arithmetic mean of its ionisation energy and electron affinity. X(A)=(1)/(2)(I.P.+E.A.) One more relationship given by him, if the values are given in eV
_easiest-way-to-calculate-electronegativity-on-pauline-scale-and-mulliken-scale.jpg)
easiest way to calculate electronegativity on Pauline scale and mulliken scale from pauling scale of electronegativity pdf Watch Video - HiFiMov.co
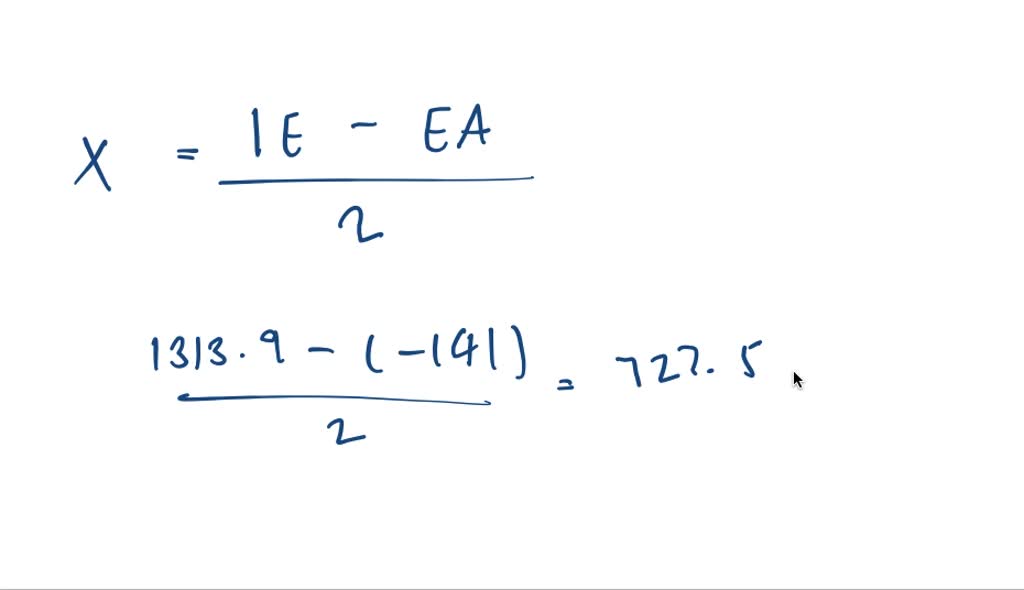
SOLVED:Using Mulliken's formula, calculate a value for the electronegativity of oxygen. Convert the result to a value on Pauling's scale. See Problem 9.149 .

WebElements Periodic Table » Periodicity » Electronegativity (Mulliken-Jaffe - sp<sup>3</sup>) » Periodic table gallery

An element has electronegativity on Mulliken Scale is 2.8 than what is the electronegativity - YouTube

Electronegativity of an element is 1.0 on the Pauling scale. Its value of Mulliken scale will be - YouTube

How to calculate electronegativity? Is there a trick for remembering the electronegativities of an element - Quora

Calculate the electronegativity value of chlorine on Mulliken's scale, given that `IP = 13.0 eV` and - YouTube

How to calculate electronegativity? Is there a trick for remembering the electronegativities of an element - Quora

If the ionisation energy and electron affinity of an element are 275 and 86 kcal mol^-1 respectively, then the electronegativity of the element on the Mulliken scale is: